OpenLab #004: Competing at an Ironman with Type 1 diabetes - Lucas (coachee) - Part I
Before we kick off today’s OpenLab, I want to express my sincere gratitude to the co-author of this edition, Lucas. Lucas is an athlete who I have been working with for an extended period of time and will compete at the Ironman in Klagenfurt this year.
He is an athlete with an incredible dedication and a keen interest in understanding the WHY behind every session. It has been an absolute pleasure to work with him and when he proposed to co-write an issue about Type 1 Diabetes in Endurance Sports (which he has been diagnosed with 18 years ago), it goes without saying that I was beyond excited in making this happen.
“As the technology improves (hybrid closed-loop pumps, faster insulin analogues, continuous glucose monitoring), managing type 1 diabetes in endurance sports has become more data-driven and safer than ever. But it remains complex. Long endurance sessions, high carbohydrate fuelling, and shifting intensities keep testing the limits of glucose regulation.
That said, type 1 diabetes should not define our athletic ceiling. It demands preparation, awareness, and strategy, but it does not limit what is possible.” — Lucas
Today’s issue covers the essential physiological background and how Type 1 Diabetes influences training and outcomes.
Part II will follow with exercise-specific glucose kinetics, fuelling strategy, practical protocols, and management limits. The essentials are distilled for a quick read in the executive summary below.




Lucas doing what he does best.
Today`s issue is a lengthy one, but this essential to explain the essential physiological background, how Type 1 Diabetes might influence training & outcomes. The essentials are distilled for a quick read in the executive summary.
Executive Summary – The Brief
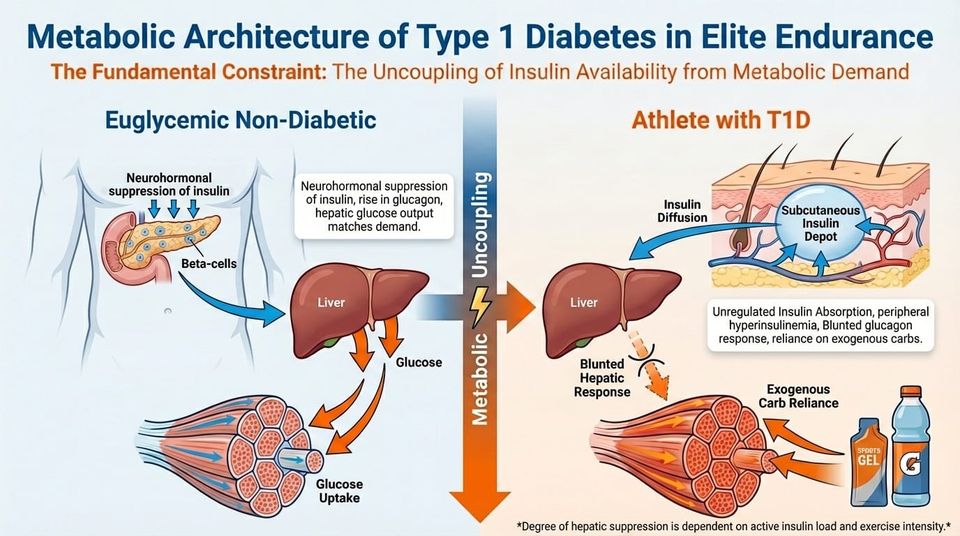
The central physiological constraint in elite endurance athletes with type 1 diabetes (T1D) is the uncoupling of insulin availability from metabolic demand. In a euglycaemic non-diabetic athlete, starting exercise triggers a rapid, neurally mediated suppression of pancreatic beta-cell insulin secretion, together with a rise in counter-regulatory hormones.
In the athlete with T1D, exogenous insulin (delivered subcutaneously via pump or injection) cannot downregulate in response to muscle contraction.
This document brings together a foundational comparison of glucose regulation in normal physiology versus T1D with a detailed look at the metabolic architecture that governs endurance performance. The key themes are:
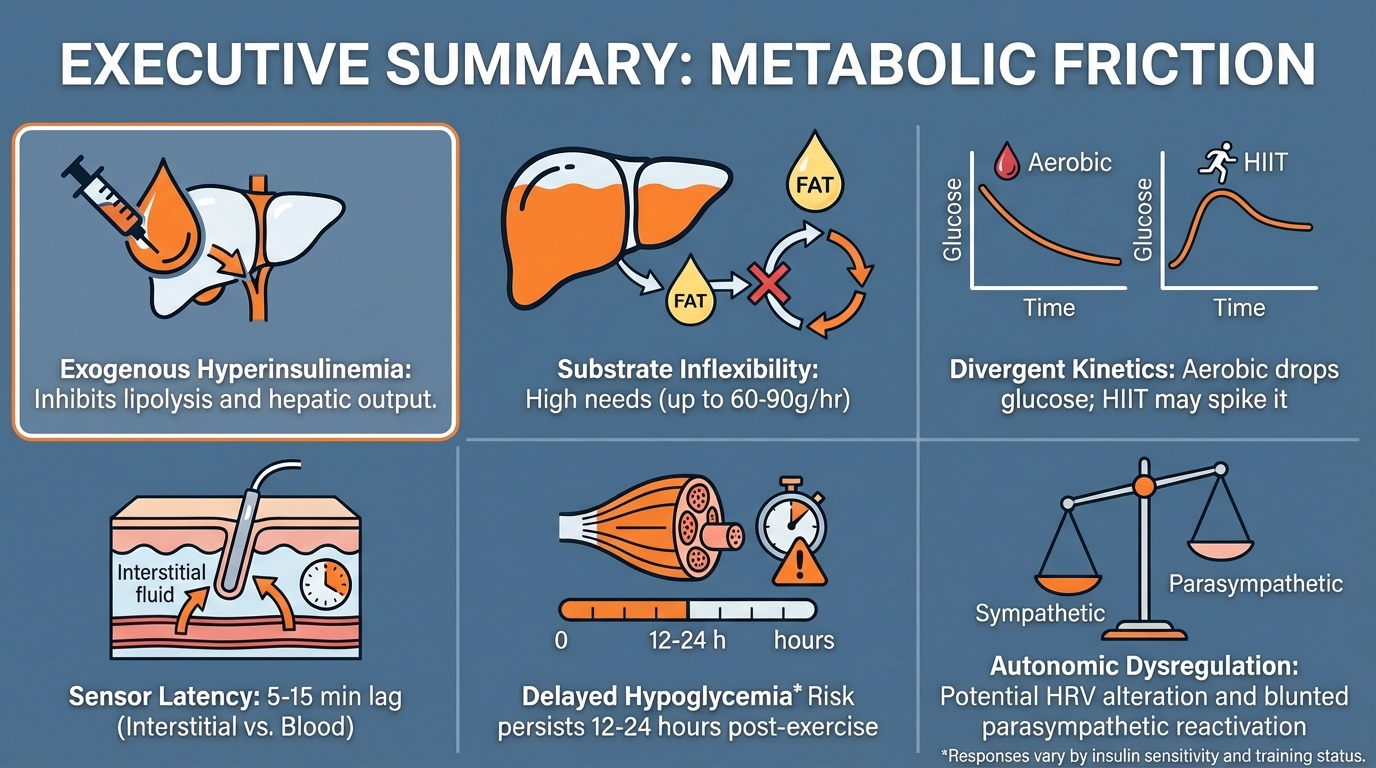
• Exogenous hyperinsulinaemia: the inability to suppress circulating insulin at exercise onset inhibits lipolysis and hepatic (from the liver) glucose output, creating high hypoglycaemia risk during aerobic effort.
• Substrate inflexibility: insulin-mediated suppression of free fatty acid mobilisation forces higher carbohydrate oxidation rates, often requiring 60–90 g CHO/hr to sustain euglycaemia.
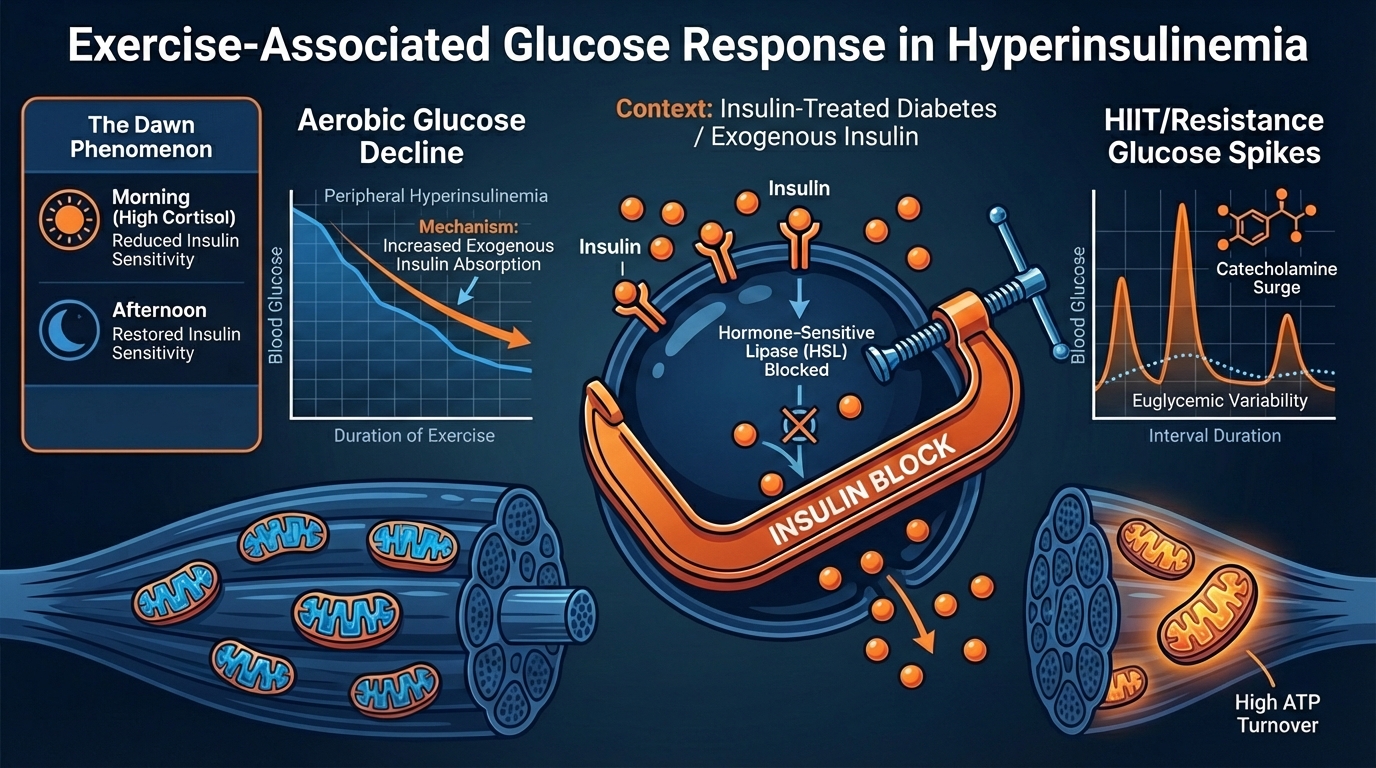
• Divergent glucose kinetics: steady-state aerobic exercise drives glucose decline, while high-intensity and anaerobic work may trigger transient hyperglycaemia through catecholamine-driven hepatic output.
• Sensor latency: CGM measures interstitial fluid with a 5–15 minute physiological lag, making predictive (rather than reactive) nutritional interventions necessary.
• Delayed hypoglycaemia: enhanced insulin sensitivity and elevated glycogen synthase activity persist for 12–24 hours post-exercise, raising the risk of nocturnal hypoglycaemia.
• Autonomic dysregulation: T1D is associated with elevated sympathetic tone and potential blunting of parasympathetic reactivation, which may alter heart rate variability and cardiovascular response profiles.
The Science at a Glance
A) Normal Physiology: A Dynamic Feedback System
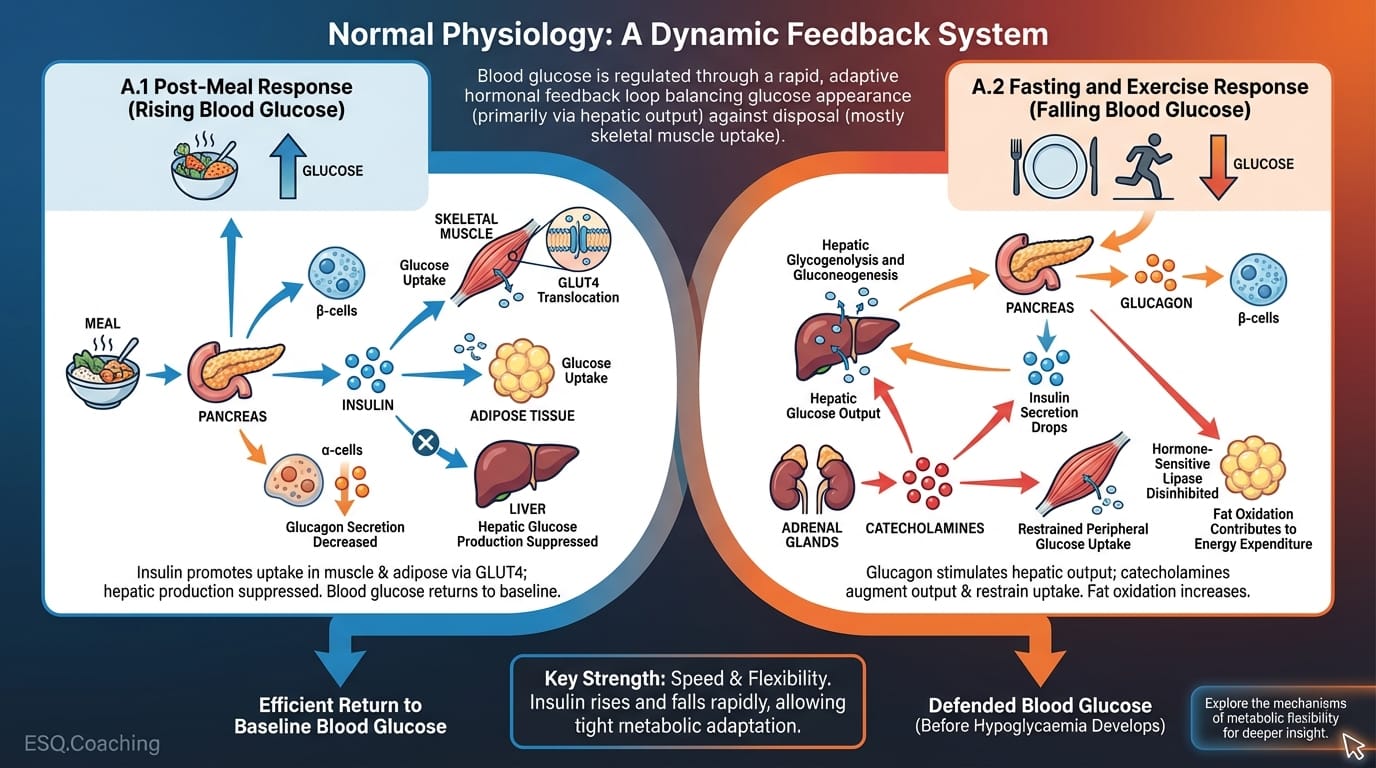
In individuals without diabetes, blood glucose is regulated through a rapid, adaptive hormonal feedback loop involving insulin (β-cells), glucagon (α-cells), catecholamines (adrenaline and noradrenaline), cortisol, and growth hormone.
This system continuously balances glucose appearance (primarily via hepatic output) against glucose disposal, which is driven mostly by skeletal muscle uptake.
A.1 Post-Meal Response (Rising Blood Glucose)
When blood glucose rises after a meal, insulin secretion increases, promoting glucose uptake in skeletal muscle and adipose tissue via GLUT4 translocation.
At the same time, hepatic glucose production is suppressed and glucagon secretion decreases. Blood glucose returns to baseline efficiently.
A.2 Fasting and Exercise Response (Falling Blood Glucose)
When blood glucose falls during fasting or exercise, insulin secretion drops within minutes. Glucagon secretion rises, stimulating hepatic glycogenolysis and gluconeogenesis.
If further defence is needed, catecholamines augment hepatic glucose output and restrain peripheral glucose uptake. Blood glucose is defended before hypoglycaemia has a chance to develop.
The key strength of this system is its speed and flexibility. Insulin can rise and fall within minutes, allowing tight metabolic adaptation during exercise. At the onset of work, falling insulin disinhibits hormone-sensitive lipase and lets fat oxidation contribute meaningfully to energy expenditure.
B) Type 1 Diabetes: Loss of Hormonal Flexibility
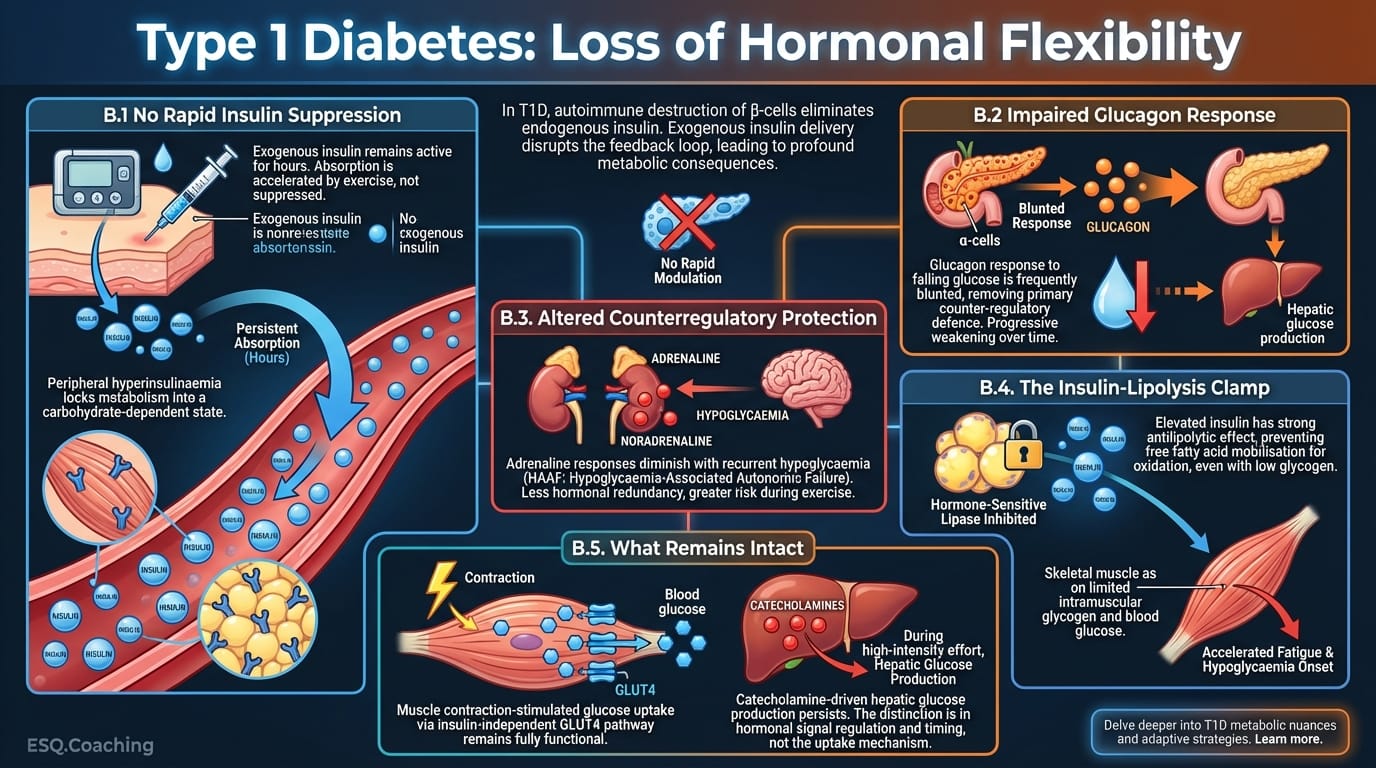
In T1D, autoimmune destruction of pancreatic β-cells wipes out endogenous insulin production. Exogenous insulin, delivered subcutaneously, cannot be modulated in response to moment-to-moment metabolic shifts. This disrupts the feedback loop in three ways.
B.1 No Rapid Insulin Suppression
Even rapid-acting insulin analogues remain active for hours after injection. Circulating insulin levels are set by absorption from subcutaneous depots, which is often accelerated (not suppressed) by increased skin blood flow and temperature during exercise.
This "peripheral hyperinsulinaemia" locks the metabolic machinery into a carbohydrate-dependent state.
B.2 Impaired Glucagon Response
The glucagon response to falling glucose is frequently blunted in T1D, removing the primary counter-regulatory defence against dropping blood glucose. Over time, this first-line protection weakens progressively.
B.3 Altered Counterregulatory Protection
Adrenaline responses may diminish with recurrent hypoglycaemia, a pattern known as hypoglycaemia-associated autonomic failure (HAAF). The result is less hormonal redundancy and greater risk during exercise.
B.4 The Insulin-Lipolysis Clamp
Elevated circulating insulin has a strong antilipolytic effect. Even with low glycogen stores, the T1D athlete may be unable to mobilise free fatty acids for oxidation effectively.
This forces skeletal muscle to rely on limited glycogen reserves and blood glucose, accelerating time to fatigue and onset of hypoglycaemia relative to non-diabetic controls.
B.5 What Remains Intact
Muscle contraction-stimulated glucose uptake via the insulin-independent GLUT4 pathway remains fully functional. Catecholamine-driven hepatic glucose production during high-intensity effort also persists. At the muscle-cell level, there is no difference between T1D and non-T1D athletes.
The distinction is in the regulation and timing of hormonal signals, not the uptake mechanism itself.
Continued in Part II — Exercise-Specific Glucose Kinetics, Fueling Strategy & Practical Protocols
Best regards and keep going full gazz,
Lucas Fosse & Dr. Thomas Mortelmans
Legal Disclaimer
The information provided in this newsletter is for educational purposes only and does not constitute medical advice, diagnosis, or treatment. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a medical condition or before starting any new fitness or nutrition regimen. The Scientist’s Notebook and ESQ Coaching are not liable for any injuries or damages that may occur from the application of the information contained herein.
References
1. Diabetes, Exercise and Competitive Sports - Gatorade Sports Science Institute
2. The competitive athlete with type 1 diabetes - PMC
3. Exercise, type 1 diabetes mellitus and blood glucose: The implications of exercise timing - PMC
4. Diabetes and Sports: Managing Your Athlete With Type 1 Diabetes - PMC
6. Exercise and Type 1 Diabetes: Must-Know Strategies for Better Control
7. Continuous Glucose Monitoring and Exercise in Type 1 Diabetes: Past, Present and Future - PMC
9. Carbohydrate Intake in the Context of Exercise in People with Type 1 Diabetes - PMC
10. Continuous Glucose Monitoring in Endurance Athletes: Interpretation and Relevance of Measurements for Improving Performance and Health - PM1. Agiostratidou, G., et al. (2017). Standardizing clinically meaningful outcome measures beyond HbA1c for type 1 diabetes. Diabetes Care, 40(12), 1622–1630.
2. American Diabetes Association. (2023). Standards of Medical Care in Diabetes, 2023. Diabetes Care, 46(Suppl. 1).
3. Bluestone, J. A., et al. (2010). Genetics, pathogenesis and clinical interventions in type 1 diabetes. Nature, 464(7293), 1293–1300.
4. Bracken, R. M., et al. (2018). Continuous glucose monitoring and exercise in type 1 diabetes: past, present and future. Biosensors, 8(3), 73.
5. Bussau, V. A., et al. (2020). The competitive athlete with type 1 diabetes. Diabetologia, 63(8), 1475–1484.
6. Cryer, P. E. (2007). Hypoglycemia in diabetes: pathophysiology, prevalence, and prevention. The American Journal of Managed Care, 13(2), 11–16.
7. DiMeglio, L. A., Evans-Molina, C., & Oram, R. A. (2018). Type 1 diabetes. The Lancet, 391(10138), 2449–2462.
8. Freckmann, G., et al. (2017). Continuous glucose profiles in healthy subjects under everyday life conditions and after different meals. Journal of Diabetes Science and Technology, 1(5), 695–703.
9. Gastaldelli, A., et al. (2000). Effect of physiological hyperinsulinemia on gluconeogenesis in non-diabetic subjects. Diabetes, 49(8), 1367–1373.
10. Horton, W. B., & Subauste, J. S. (2016). Diabetes and sports: managing your athlete with type 1 diabetes. Current Sports Medicine Reports, 15(5), 310–320.
11. Moser, O., et al. (2020). Carbohydrate intake in the context of exercise in people with type 1 diabetes. Nutrients, 12(10), 3159.
12. Munzel, T., et al. (2024). Continuous glucose monitoring in endurance athletes: interpretation and relevance of measurements for improving performance and health. Sports Medicine - Open, 10, 17.
13. Riddell, M. C., et al. (2017). Exercise management in type 1 diabetes: a consensus statement. The Lancet Diabetes & Endocrinology, 5(5), 377–390.
14. Scott, S., & Helleputte, S. (2025). Empowered: The Science of Exercise with Type 1 Diabetes. Sestante Analytics Ltd.
15. Sherwin, R. S., & Sacca, L. (1984). Effect of epinephrine on glucose metabolism in humans. American Journal of Physiology, 247(2), E157–E165.
16. Szmigiel, L., et al. (2013). Sweet 452 km: a report on the first type 1 diabetes patient to finish Double Ironman. Experimental and Clinical Endocrinology & Diabetes, 121(7), 421–424.
17. Yardley, J. E., et al. (2022). Exercise, type 1 diabetes mellitus and blood glucose: the implications of exercise timing. Diabetes & Metabolic Syndrome: Clinical Research & Reviews, 16(10), 102620.
18. Zaharieva, D. P., & Riddell, M. C. (2018). 24 days to Olympic Games: managing a competitive athlete with type 1 diabetes. Diabetes Care, 1(1), 143–147.
Found this useful?
Get articles like this and free training calculators in your inbox every week.
Recommended reads
If this was useful, these are the other research-driven newsletters I actually read.
Member discussion